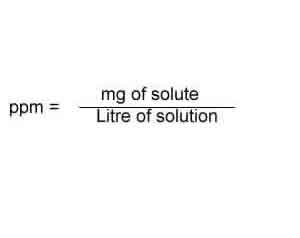
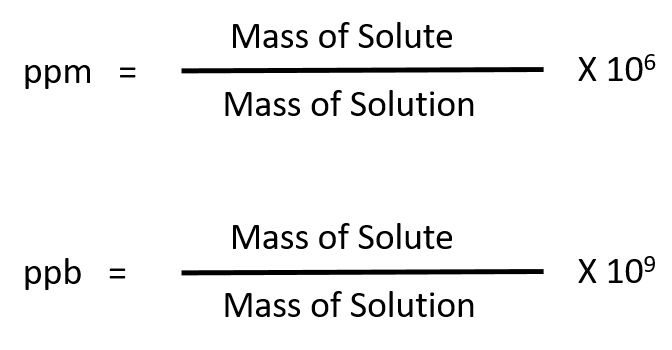
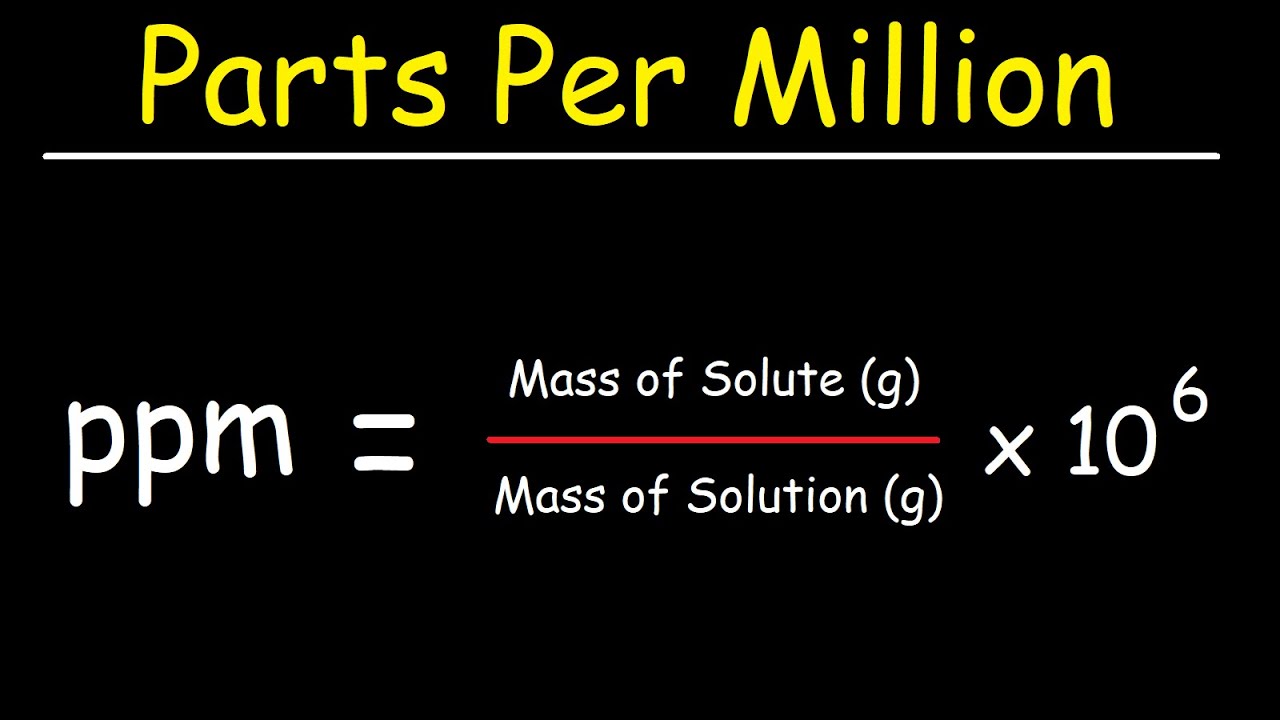SOLVED: Explain how to prepare standard solutions (5 ml each) of NaCl with the following concentrations: 10, 20, 30, 50, 70 ppm. The stock solution has a concentration of 0.05% w/v. (5 marks)

PPM solution | How to prepare 5 ppm, 10 ppm, 15ppm, 20ppm, 40ppm,1000ppm solutions! Stock Solution - YouTube